NSW Biobank Certification Program
The NSW Health Statewide Biobanking Framework aims to create a world-class, sustainable approach to managing human biobanks, the specimens they house and improve integration between research, diagnostics and clinical care. NSW Biobank Certification Program forms part of this framework.
The voluntary certification scheme aims to improve and harmonise biobanking standards through education and communication of best practices and Standard Operating Procedures (SOPs). It is based on the certification program designed and delivered in Canada by the Office of Biobank Education and Research (OBER) at the University of British Columbia (UBC) in Vancouver and the Canadian Tissue Repository Network (CTRNet). The NSW Health Pathology program contains Australian-specific content regarding the laws, regulations and guidelines applicable to research and information about both private and public health care. The evaluation component of the education was designed in conjunction with My Health Learning which supports education and training for excellent health care across the NSW Health system.
Online education modules are key to the program. The education modules address all aspects of biobanking and focus on rationale underlying standards. Our certification program is applicable to all types of biobanks and pathology laboratories that engage in biobanking and research activities such as clinical trials. Upon completion of the program, the certificates are endorsed by representatives from both the University of British Columbia and NSW Health Pathology.
What are the goals of the program?
- To promote adherence with best practices for biobanking, to achieve high and comparable quality across biobanks
- To provide educational resources for new and existing biobanks and pathology laboratories engaged in biobanking activities to increase and evaluate knowledge
- To facilitate the adoption of best practice standards
- To foster both public and researcher confidence in biobanks
What is unique about the program?
- Focused on education
- Developed by biobank experts for biobanks
- Applicable to all types of biobanks (including research studies with biobanking components) and pathology laboratories engaged in biobanking activities
- Adapted to meet the specific setting of Australian biobanks
- Low cost
The Certification Process
The program is comprised of two linked phases: Registration and Certification (see Fig. 1)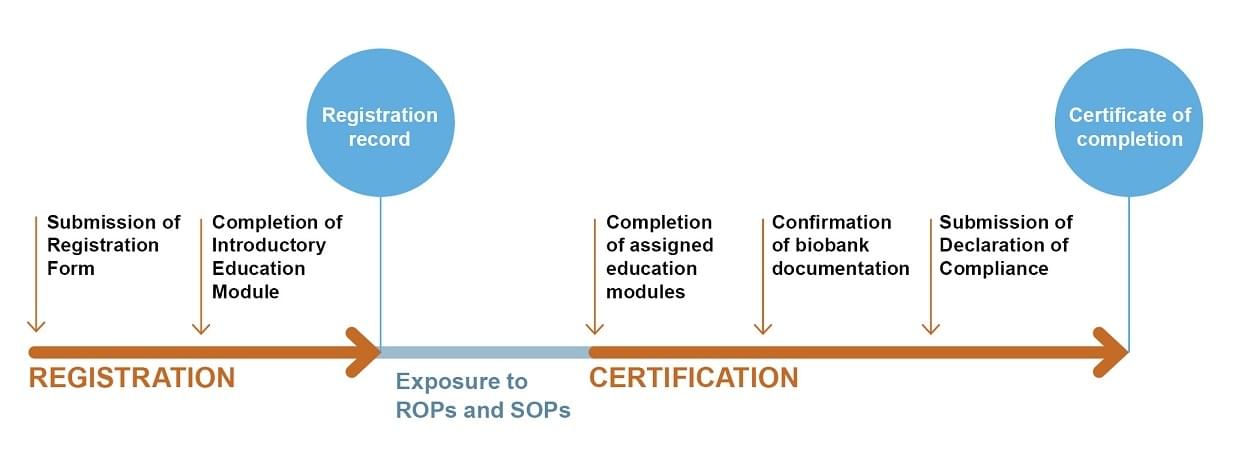
Steps explained
Registration:This is the first phase of the program. The goal of this phase is to collect basic information about the biobank/pathology laboratory and complete an introductory education course that covers all the basic aspects of biobanking.
The Biobank/Pathology Lab Team Leader (e.g. the biobank principal investigator, biobank/laboratory manager, pathologist or most responsible person. This task may be delegated to a less senior staff member.):
- Creates an account and completes the Registration Form.
- Completes the online introductory overview education module Basics of Biobanking including passing the HETI test with a score of >80% at the end of the module.
- Receives a record of registration and education module completion report.
- Receives access to the best practice standards documents and shares these with their biobank/pathology laboratory team members.
The estimated time to completion is 2 - 3 hours.
Certification:This is the second phase of the program. The goals of this phase are to provide additional education modules based on the biobank/laboratory type and confirm that the biobank/laboratory has key documents to determine whether or not the biobank/laboratory has adapted to best practice standards.
The biobank leader should check the costs involved in certifying (see certification for more information). Once the biobank leader has clicked to begin certification, they will be able to immediately continue through the certification pathway. The biobank leader will be emailed an invoice from NSWHP at a later date. Failure to pay the invoice will result in restriction of program content until payment is received.
The Biobank/Pathology Lab Team Leader (e.g. the biobank principal investigator, biobank/laboratory manager, pathologist or most responsible person. This task may be delegated to a less senior staff member.):
- Invites their biobank/lab team members to create accounts.
- Assigns relevant education modules to biobank/lab team members.
- Completes required education module(s) including passing the HETI test(s) at the end of each module with a score of >80%.
- Completes a checklist of existing key biobank/lab documentation.
- Ensures biobank/lab team members have completed required education modules.
- Uploads documents for review by the Biobank Certification Officer.
- Submits the biobank's Declaration of Compliance with best practices.
- Receives a completion of certification and education module completion reports.
- Clicks on the link in the invitation email to create an account.
- Completes the online introductory overview education module Basics of Biobanking.
- Completes the education modules recommended by the biobank/lab leader.
- Passes the HETI test(s) following each education module with a score of >80%.
The effort required to complete certification will vary depending on the biobank/pathology lab operational complexity and number of personnel. Most biobanks/labs will require 12 hours over one to three months of intermittent activity spread between various personnel.
Education OnlySome users, such as members of a human research ethics committee, may wish to access the education modules without registering or certifying a biobank or pathology laboratory. This is possible by selecting ‘Education Only’ from the ‘Enrol’ tab.

